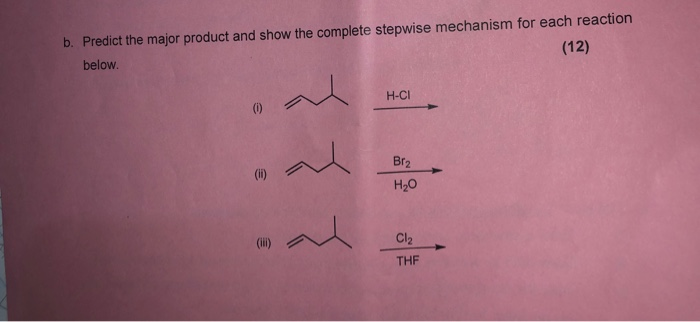
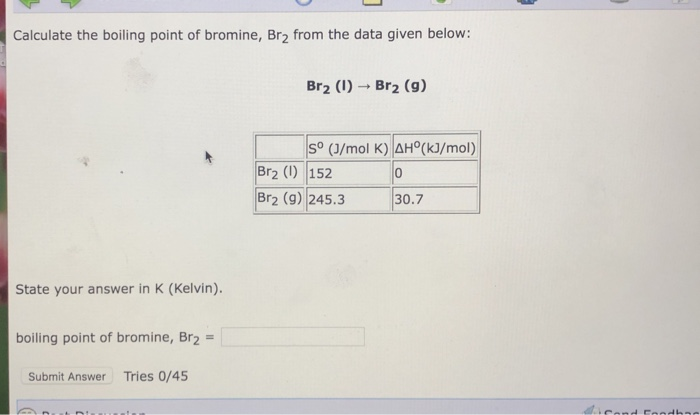
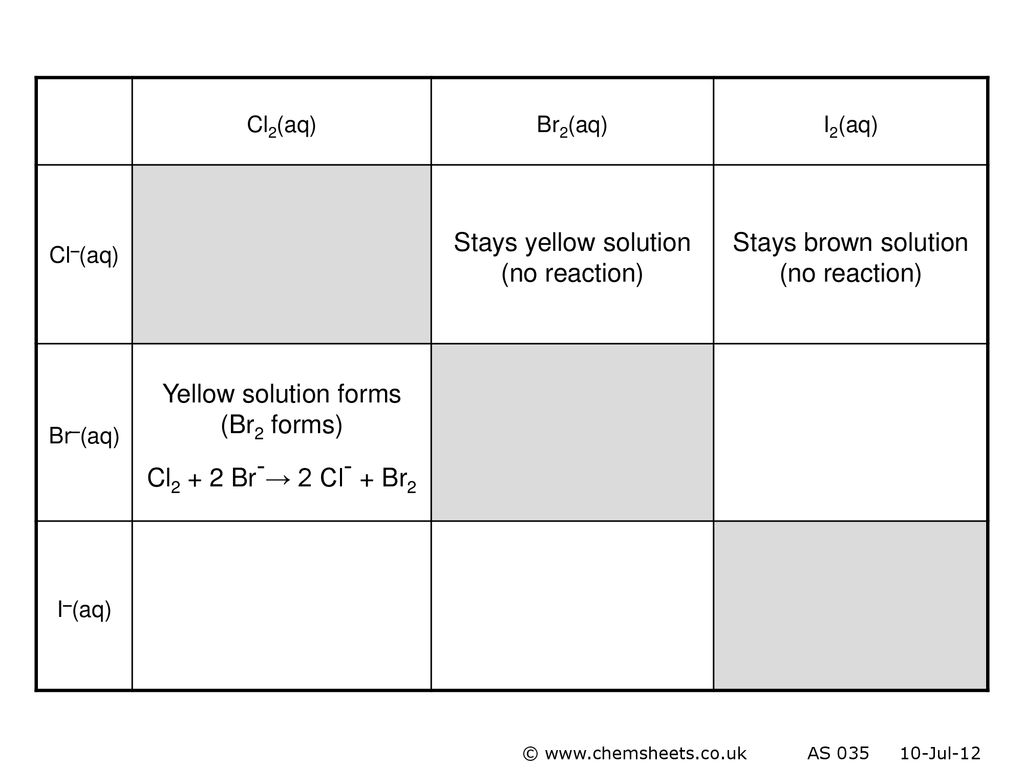
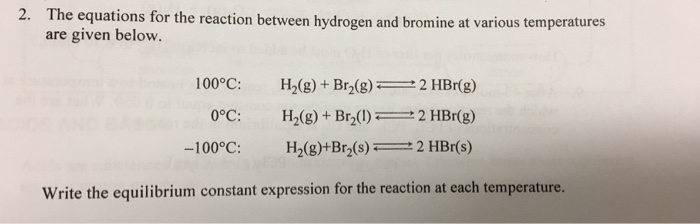Q5 from 5.3 Identify all the species in the table which could convert I–aq) into I2(aq) but which could not convert Br–(aq) into Br2(l). - ppt video online download
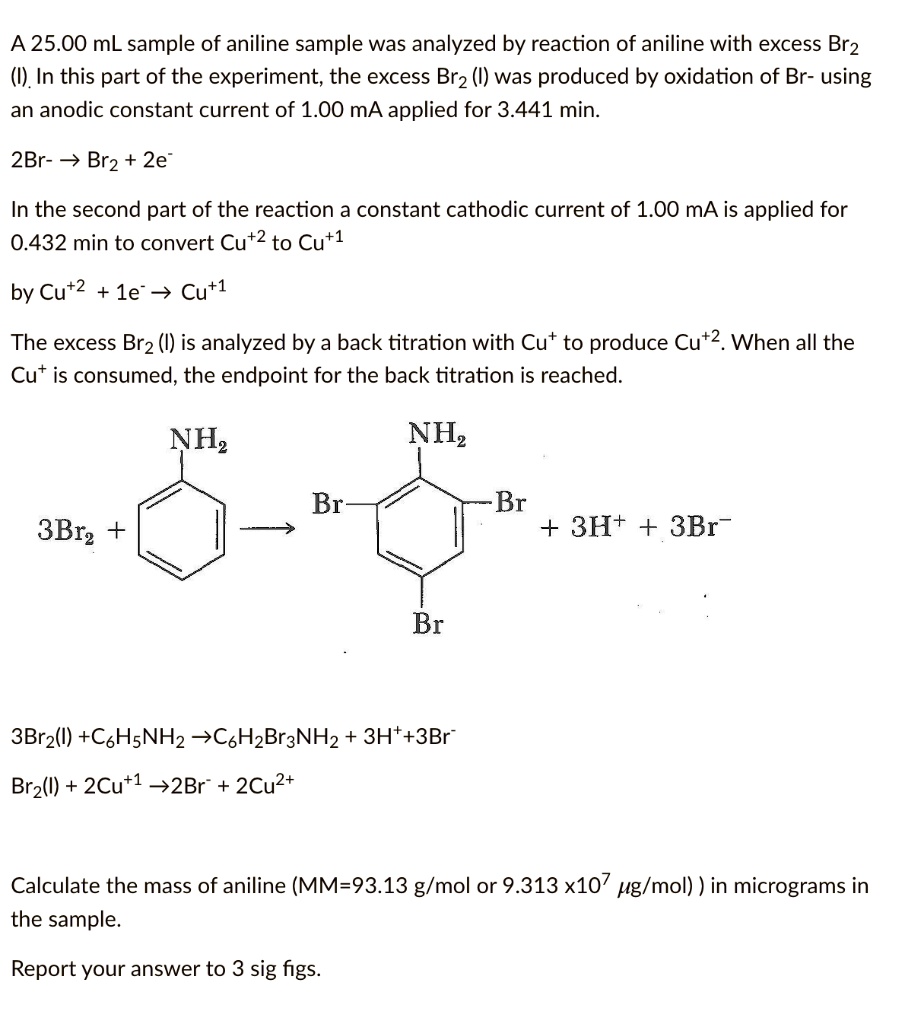
SOLVED: A 25.00 mL sample of aniline sample was analyzed by reaction of aniline with excess Br2 (I). In this part of the experiment; the excess Brz (I) was produced by oxidation

SOLVED: determine the substance reduced this redox reaction 2 NaBr (s) + Cl2 (g) —> Br2 (I) + 2 NaCI (s) NaBr, Na, Cl2, NaCl, Br


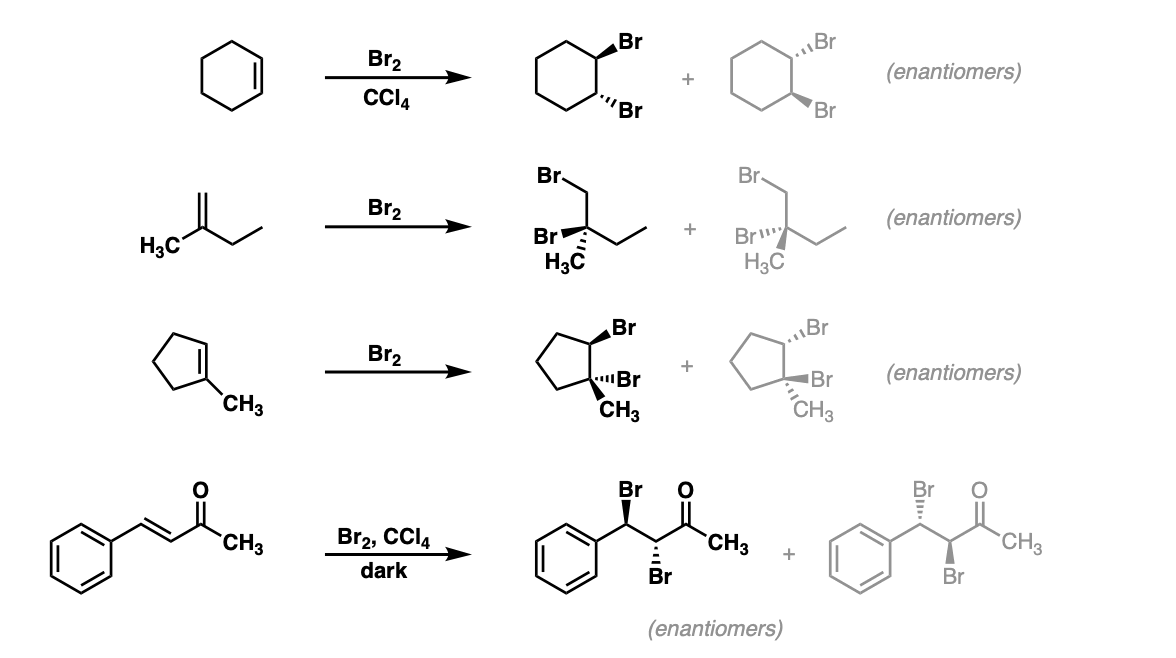




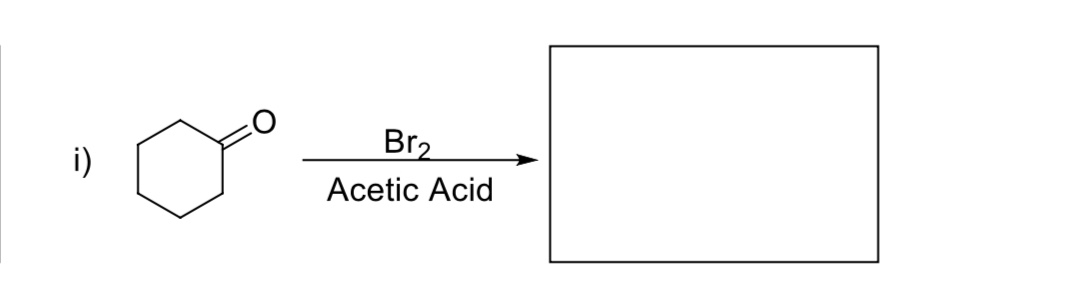


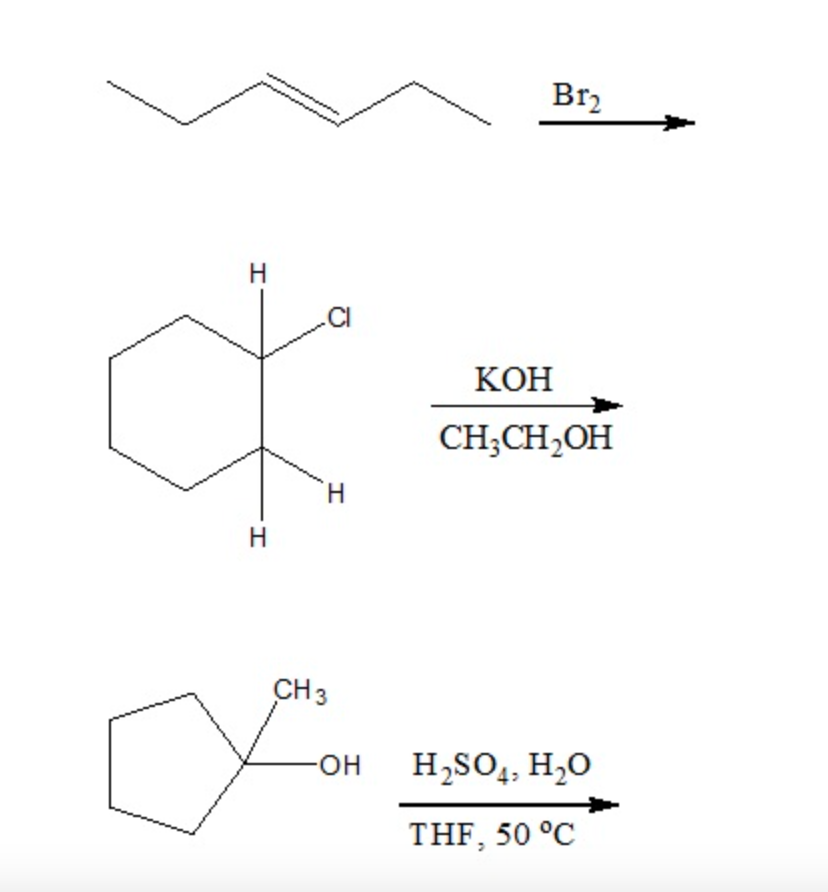



![Bromohydrin formation [Br2/H2O] - ChemistryScore Bromohydrin formation [Br2/H2O] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Bromohydrin-formation1-768x312.png)