
SOLVED: When solid phosphorus, P, reacts with gaseous chlorine, Cl2, it forms liquid phosphorus trichloride, PCl3. When 4.62 grams of phosphorus reacts with excess chlorine at constant pressure 47.8 kJ of heat

Gaseous NOCl is placed in a closed container at 173 oC, where it partially decomposes to NO and Cl2:2 NOCl(g) 2 NO(g) + 1 Cl2(g) At equilibrium it is found that p(NOCl) =

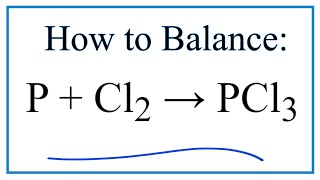
How to balance P + Cl2 = PCl5|Chemical equation P + Cl2 = PCl5|reaction balance P + Cl2 = PCl5 - YouTube

P+Cl₂=PCl₅ Balanced Eq.| phosphorus+ chlorine gas to form Phosphorus pentachloride Balanced Equation - YouTube | Balance, Phosphorus, Equation
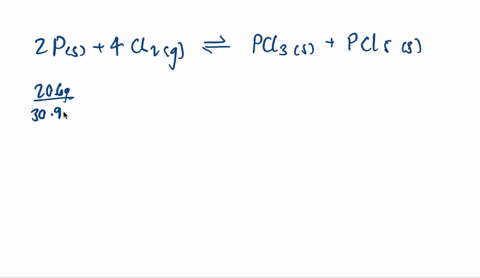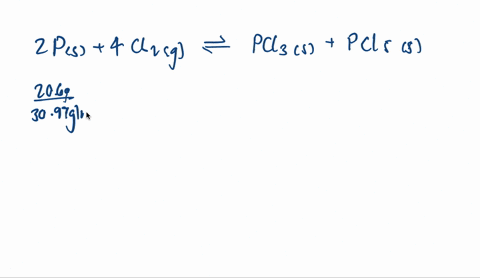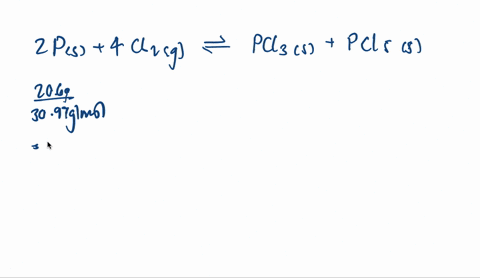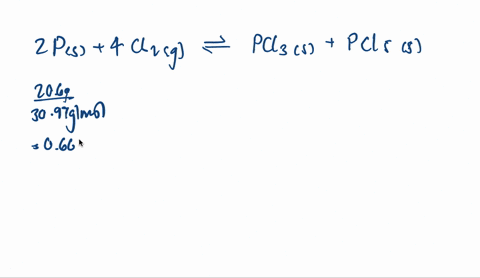
SOLVED: A mixture of 20.6 g of P and 79.4 g Cl2 reacts completely to form PCl3 and PCl5, which are the only products. Determine the mass of PCl3 that forms.

At certain temperature(T) for the gas phase reaction: 2H2O(g) + 2Cl2(g) 4HCl(g) + O2(g) , Kp = 12 × 10^8 atm. If Cl2, HCI & O2 are mixed in such a manner
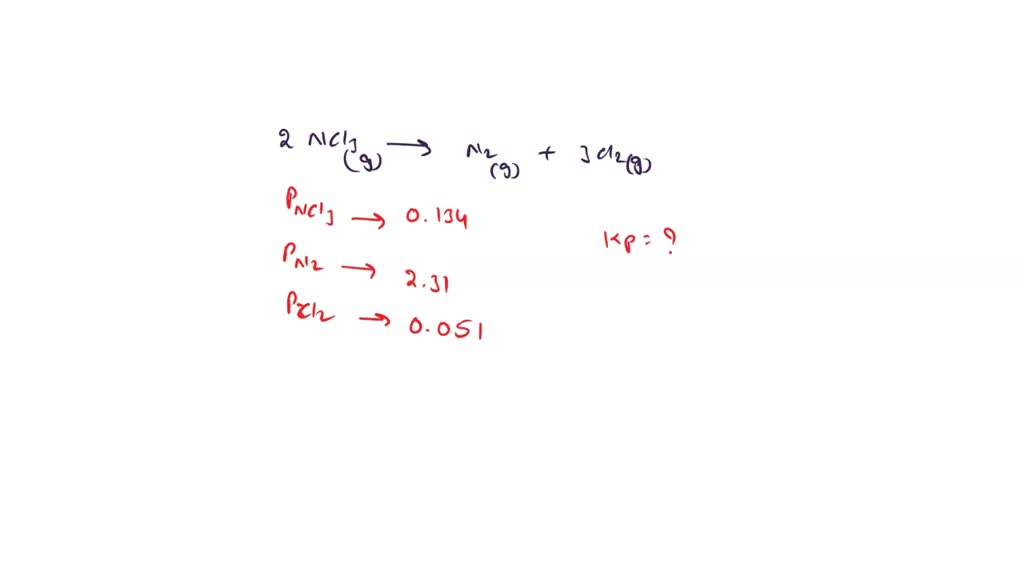
SOLVED: For the reaction 2NCl3(g) ↔ N2(g) + 3Cl2(g), the equilibrium pressures are P(NCl3) = 0.134 atm P(N2) = 2.31 atm P(Cl2) = 0.0510 atm Determine Kp for this reaction

OneClass: Chem 401 Discussion workshop #5 Name 3. Initially, ph-pCl2=0.10 atm plci-0 a) Set up expres...

Model systems for a stepwise hydrolysis of (a) PCl3, (b) P(OH)Cl2, (c)... | Download Scientific Diagram
CH3CH2 COOH,+Cl2(red P)→ A ,(alc.KOH) → B. What is B? - Sarthaks eConnect | Largest Online Education Community
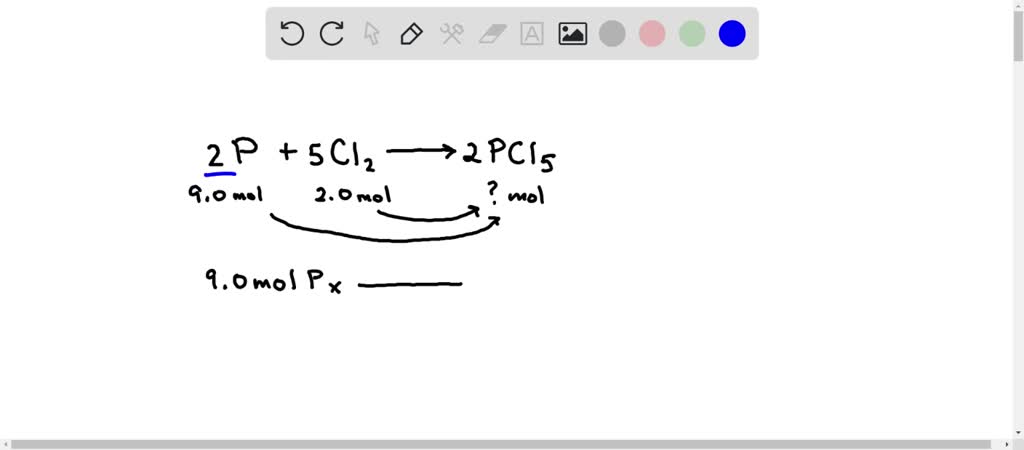
SOLVED: Solid phosphorus and chlorine gas react to form solid phosphorus pentachloride. Suppose you have 9.0 mol of P and 2.0 of Cl2 in a reactor. Calculate the largest amount of PCl5

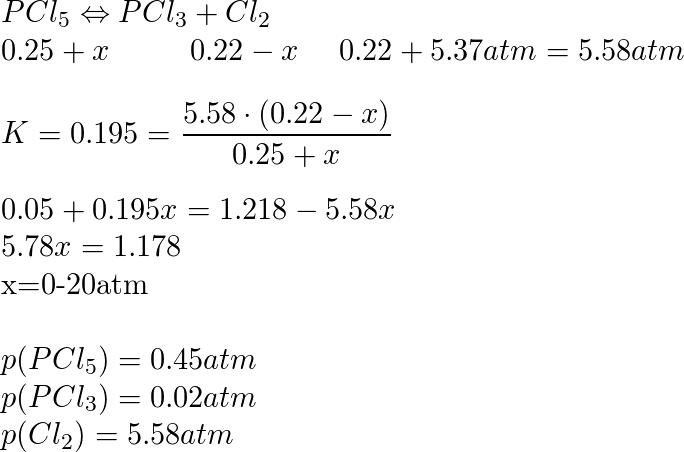

![Class 12] An organic compound (A) with molecular formula C2Cl3O2H is Class 12] An organic compound (A) with molecular formula C2Cl3O2H is](https://d1avenlh0i1xmr.cloudfront.net/b998b889-9a60-4ab5-b89b-e9db2209b7a9/question-34-choice-b---an-organic-compound-(a)-with-molecular-formula---teachoo.jpg)




![In the equation CH3COOH + Cl2 [ - HCl]Red P A , the compound A is : In the equation CH3COOH + Cl2 [ - HCl]Red P A , the compound A is :](https://d1hhj0t1vdqi7c.cloudfront.net/v1/aG5Wc3JqMnNnVGs=/sd/)

